Molar mass: The mass of a mole of a substance.Limiting reagent: The reactant which will determine the theoretical maximum amount of product formed. 
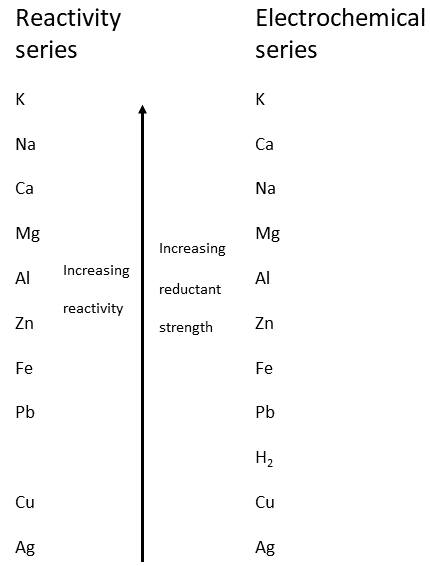
Ion: An atom or molecules where the total number of protons is not equal to the total number of electrons.Homogeneous mixtures: which have both uniform composition and uniform properties throughout the mixture.Heterogeneous mixtures: which have a non-uniform compositions and hence their properties vary throughout the mixture.Formula, structural: Shows the arrangement of atoms and bonds within a molecule.Formula mass: The mass of one formula unit of that substance.Formula, empirical: The formula obtained by experiment, showing the simplest whole number ratio of atoms of each element in a particle of the substance.Excess: The reactant which there is more of than needed to react with all of the limiting reagent.Element:Ěn element contains atoms of only one type.Concentration: a measure of the amount of dissolved substance contained per unit of volume.Compound: Two or more types of atoms chemically bonded together.Chemical reaction:Ě reaction in which bonds in the reactants are broken and bonds in the products are formed and vice versa resulting in an energy change between the reacting system and its surroundings.Avogadro’s constant: The number of particles in 12g of 12C.Ė.022 x 10^23.Absolute Zero is zero on the Kelvin scale, 0 K (on the Celsius scale this is -273 ☌).1.10 Topic 10 and 20 – Organic ChemistryĬore Topic 1 - Stoichiometry.1.9 Topic 9 and 19 – Oxidation and Reduction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
